For a long time, the process of communication between nerves and their target tissues was largely unknown to physiologists. With the development of electrophysiology and the discovery of the electrical activity of neurons, it was discovered that signal transmission from neurons to their target tissues is mediated by action potentials. An action potential is defined as a sudden, rapid, transient, and propagating change in the resting membrane potential. Only neurons and muscle cells are capable of generating an action potential; that property is called excitability.
Definition
Action potentials are nerve signals. Neurons generate and conduct these signals throughout their processes to transmit them to target tissues. Upon stimulation, they will be stimulated, inhibited, or modulated in some way.
Steps
But what causes the action potential? From an electrical aspect, it is caused by a stimulus with a certain value expressed in millivolts [mV]. Not all stimuli can cause an action potential. The appropriate stimulus must have sufficient electrical value to reduce the negativity of the nerve cell to the threshold of the action potential. Thus, there are subthreshold, threshold and suprathreshold stimuli.
- Subthreshold stimuli cannot cause an action potential.
- Threshold stimuli have enough energy or potential to produce an action potential (nerve impulse).
- Suprathreshold stimuli also produce an action potential, but its strength is greater than that of threshold stimuli.
- So, an action potential is generated when a stimulus changes the membrane potential to threshold potential values. The threshold potential is usually between -50 and -55 mV. It is important to know that the action potential behaves according to the law of all or nothing. This means that any subthreshold stimulus will not cause anything, whereas threshold and suprathreshold stimuli produce a full response from the excitable cell.
Is an action potential different depending on whether it is caused by a threshold or suprathreshold potential? The answer is no. The length and amplitude of an action potential are always the same. However, increasing the strength of the stimulus causes an increase in the frequency of an action potential. An action potential propagates along with the nerve fibre without diminishing or weakening its amplitude and length. Also, after an action potential is generated, neurons become refractory to stimuli for a certain period of time where they cannot generate another action potential.
Stages
From the point of view of ions, an action potential is caused by temporary changes in the permeability of the membrane for diffusible ions. These changes cause the ion channels to open and the ions to decrease their concentration gradients. The value of the threshold potential depends on the permeability of the membrane, the intracellular and extracellular concentration of ions, and the properties of the cell membrane.
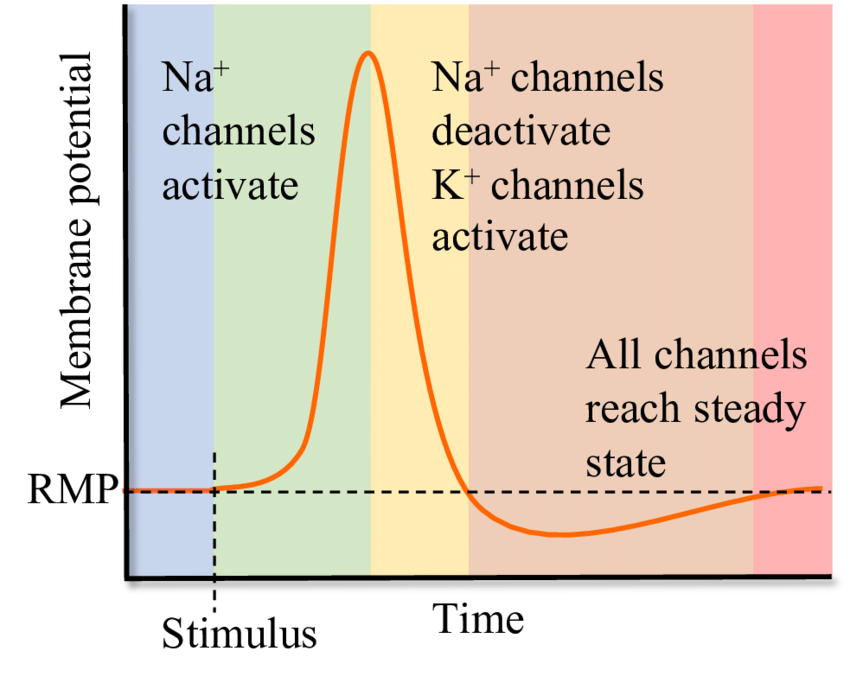
An action potential has three phases: depolarization, overshoot, and repolarization. There are two more states of the membrane potential related to the action potential. The first is hyperpolarization that precedes depolarization, while the second is hyperpolarization that follows repolarization.
Hyperpolarization is the initial increase in membrane potential to the value of the threshold potential. The threshold potential opens voltage-gated sodium channels and causes a large influx of sodium ions. This phase is called depolarization. During depolarization, the interior of the cell becomes increasingly electropositive, until the potential approaches the electrochemical equilibrium for sodium of +61 mV. This phase of extreme positivity is the overdrive phase.
After the overshoot, the permeability to sodium suddenly decreases due to the closure of its channels. The overshoot value of the cell potential opens voltage-gated potassium channels, causing a large outflow of potassium, which lowers the electropositivity of the cell. This phase is the repolarization phase, the purpose of which is to restore the resting membrane potential. Repolarization always leads first to hyperpolarization, a state in which the membrane potential is more negative than the default membrane potential. But soon after, the membrane re-establishes the values of the membrane potential.
After reviewing the functions of the ions, we can now define threshold potential more precisely as the value of the membrane potential at which voltage-gated sodium channels open. In excitable tissues, the threshold potential is about 10 to 15 mV lower than the resting membrane potential.

Refractory period
The refractory period is the time after an action potential is generated, during which the excitable cell cannot produce another action potential. There are two subphases of this period, absolute and relative refractoriness.
1. Absolute refractoriness overlaps depolarization and about 2/3 of the repolarization phase. A new action potential cannot be generated during depolarization because all voltage-gated sodium channels are already open or are opening at their maximum rate. During early repolarization, a new action potential is impossible since the sodium channels are inactive and need the resting potential to be in a closed state, from which they can return to an open state. Absolute refractoriness ends when enough sodium channels recover from their inactive state.
2. Relative refractoriness is the period in which the generation of a new action potential is possible, but only in response to a suprathreshold stimulus. This period overlaps the final 1/3 of repolarization.
Propagation of the action potential.
An action potential is generated in the body of the neuron and propagates down its axon. Propagation does not diminish or affect the quality of the action potential in any way, so the target tissue receives the same impulse no matter how far away it is from the cell body.
The action potential is generated at a point on the cell membrane. It spreads across the membrane, and each succeeding part of the membrane depolarizes sequentially. This means that the action potential does not move but instead elicits a new action potential from the adjacent segment of the neuronal membrane.
We need to emphasize that the action potential always propagates forward, never backward. This is due to the refractoriness of the parts of the membrane that were already depolarized, so the only possible direction of propagation is forward. Because of this, an action potential always propagates from the cell body, down the axon to the target tissue.
The speed of propagation depends largely on the thickness of the axon and whether or not it is myelinated. The larger the diameter, the higher the rate of propagation. Propagation is also faster if an axon is myelinated. Myelin increases the speed of propagation because it increases the thickness of the fibre. Furthermore, myelin allows for saltatory conduction of the action potential, since only the nodes of Ranvier are depolarized and myelin nodes are skipped. In unmyelinated fibres, each part of the axonal membrane must undergo depolarization, which makes propagation significantly slower.
Synapse
A synapse is a junction between the nerve cell and its target tissue. In humans, synapses are chemical, meaning that the nerve impulse is transmitted from the axon terminal to the target tissue by chemicals called neurotransmitters (ligands). If a neurotransmitter stimulates the action of the target cell, then it is an excitatory neurotransmitter. On the other hand, if it inhibits the target cell, it is an inhibitory neurotransmitter. Depending on the type of target tissue, there are central and peripheral synapses. Central synapses occur between two neurons in the central nervous system, while peripheral synapses occur between a neuron and muscle fibre, peripheral nerve, or gland.
Each synapse consists of:
- Presynaptic membrane: membrane of the terminal button of the nerve fibre.
- Postsynaptic membrane: membrane of the target cell.
- Synaptic cleft: a gap between the presynaptic and postsynaptic membranes
Numerous vesicles containing neurotransmitters are produced and stored within the terminal button of the nerve fibre. When the presynaptic membrane is depolarized by an action potential, voltage-gated calcium channels open. This leads to an influx of calcium, which changes the state of certain membrane proteins in the presynaptic membrane and results in the exocytosis of the neurotransmitter in the synaptic cleft.
The postsynaptic membrane contains receptors for neurotransmitters. Once the neurotransmitter binds to the receptor, ligand-gated channels in the postsynaptic membrane open or close. These ligand-gated channels are the ion channels and their opening or closing will cause a redistribution of ions in the postsynaptic cell. Depending on whether the neurotransmitter is excitatory or inhibitory, this will result in different responses.